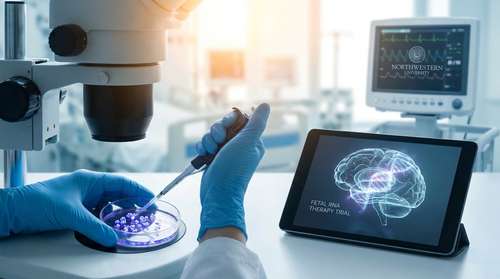
A devastating diagnosis of KCNT1-related epilepsy usually means a newborn will suffer up to 100 life-altering seizures a day almost immediately after birth. By the time medical teams begin managing symptoms, critical damage to the infant’s developing brain has often already occurred. However, a groundbreaking discovery from a late April 2026 study published in Nature Communications is drastically shifting this paradigm. Scientists have successfully demonstrated that an in utero epilepsy treatment could intercept this rare neurological condition before a child takes their first breath. By intervening during the middle of pregnancy, this novel therapy aims to preserve early brain plasticity and permanently alter the trajectory of a child's life.
Northwestern University Pediatric Research Unlocks Early Intervention
The question of exactly when to administer therapeutics to outpace genetic diseases has long frustrated medical professionals. According to the latest Northwestern University pediatric research, the answer might be much earlier than previously thought. Richard Smith, an assistant professor of pharmacology and pediatrics at the Northwestern University Feinberg School of Medicine, recently led a research team to investigate the timeline of early brain development and neurological damage.
Their findings revealed that abnormal electrical currents begin wreaking havoc on fetal brain cells long before a baby is born. The researchers discovered that the early brain is an amazingly plastic structure. Missing the earliest therapeutic window makes reversing neurological harm significantly more difficult later on as clinicians struggle to manage symptoms in patients. By targeting the perinatal period, specialists can theoretically prevent the hyper-excitation that causes long-term cognitive and physical deficits, setting a new gold standard for proactive medicine.
Targeting the KCNT1 Gene Mutation with Fetal RNA Therapy
To test their clinical hypothesis, the scientific team focused on a highly severe, treatment-resistant disorder driven by a KCNT1 gene mutation. Children born with this specific genetic anomaly overproduce excessive electrical activity in their neurons, leading to relentless seizures and, tragically, premature death in many cases. The standard anticonvulsant medications available today simply cannot keep up with the aggressive nature of the disease.
Rather than relying on traditional pharmaceuticals, the laboratory utilized an experimental fetal RNA therapy. When researchers applied this specialized treatment to patient-derived excitatory neurons grown in the lab, they witnessed a dramatic reduction in abnormal electrical currents. The overactive cells essentially cooled down, returning to a normal baseline of activity.
The Mechanics of Antisense Oligonucleotides
The secret behind this success lies in a technology known as an antisense oligonucleotide (ASO). These are synthetic strings of nucleic acids designed to bind to specific messenger RNA molecules. In the context of the genetic mutation, the ASO intercepts the faulty instructions that tell the brain cells to over-fire and reduces KCNT1 activity. By silencing this dangerous command, the therapy effectively normalizes the neural network's electrical activity without permanently altering the underlying DNA structure.
Clinical Success at 15 Weeks Gestation
What makes this study truly revolutionary is the timeline. The research team tested the RNA-based therapy on developing human brain cells equivalent to the middle of pregnancy, specifically 15 to 21 weeks gestation. At this early stage, the therapy successfully suppressed the excessive neuronal firing. Proving that an in utero epilepsy treatment works at this specific developmental milestone establishes a key clinical basis for targeting the perinatal period before the fetal brain fully forms.
Navigating the Path to Clinical Trials
While the laboratory results are undeniably promising, transitioning an in utero epilepsy treatment from isolated cell cultures to living human patients involves rigorous safety protocols. Delivering a fetal RNA therapy requires incredible precision to ensure the molecules reach the fetal brain barrier without causing distress to the mother or child. Researchers are focused on understanding how to strengthen this cooling-off process in living brains where neurons fire in highly complex patterns. This critical next phase of testing will help establish the exact methods required to neutralize the KCNT1 gene mutation safely before wide-scale clinical trials can begin.
The True Impact of Preventing Childhood Seizures
For families carrying genetic markers for severe neurodevelopmental disorders, the emotional toll of anticipating a profoundly sick newborn is immense. The primary goal of modern prenatal neurological care is rapidly evolving from simply diagnosing structural anomalies via ultrasound to actively repairing cellular defects directly in the womb.
Preventing childhood seizures through early ASO therapy means an infant's brain can develop on a normal timeline without the constant, destructive interruption of electrical storms. In practice, protecting the delicate fetal brain from hyper-excitation preserves basic functional milestones. Skills like walking, talking, and social interaction—abilities that children with KCNT1-associated epilepsy typically lose—can be protected. The therapy effectively turns off the genetic switch that causes the potassium channels to malfunction.
Shaping the Future of Pediatric Medicine 2026 and Beyond
We are witnessing a profound shift in maternal-fetal medicine. While the current breakthrough specifically addresses a single devastating mutation, the clinical framework established by the Northwestern laboratory offers hope for a vast array of other genetic neurological conditions. If specialists can safely deliver these targeted RNA therapies to fetuses or extremely premature infants, the ripple effects across the entire healthcare landscape will be massive.
The future of pediatric medicine 2026 is clearly moving toward proactive, highly targeted treatments. By identifying genetic anomalies early in pregnancy and intervening at the precise developmental moment, medical professionals are no longer just managing catastrophic symptoms after the fact. They are actively rewriting the patient's biological destiny, ensuring that a rare genetic trait does not have to result in a lifetime of suffering.