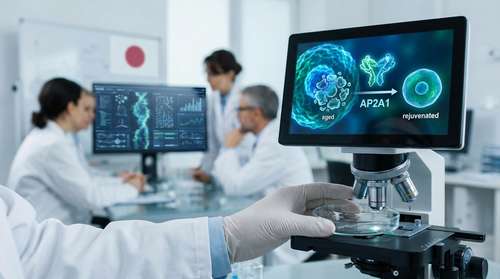
In a groundbreaking development that could fundamentally change how we understand human longevity, researchers at Osaka University have identified a critical "molecular switch" capable of reversing cellular aging. The discovery, announced this week, centers on the AP2A1 protein aging switch, a mechanism that appears to control the transition between youthful cellular function and senescence. By manipulating this protein, scientists successfully reverted "zombie" cells to a healthy, active state, marking a significant leap forward in the quest to reverse biological aging in 2026.
The "Off Switch" for Cellular Senescence
For decades, scientists have hunted for the drivers behind cellular senescence—the state where cells stop dividing and accumulate, spewing toxic inflammatory signals (SASP) that degrade surrounding tissue. Until now, the structural maintenance of these enlarged, dysfunctional cells remained a mystery. The team from Osaka University, led by Professor Shinji Deguchi and researcher Pirawan Chantachotikul, found the answer in an unexpected place: the cytoskeleton.
The study reveals that the protein AP2A1 (Adaptor Protein Complex 2, Alpha 1 Subunit) is not merely a bystander but a driver of the aging process. In senescent cells, AP2A1 levels skyrocket, specifically along "stress fibers"—the cables that hold a cell's shape. This accumulation locks the cell into a rigid, enlarged, and inflammatory state. When the researchers flipped this "switch" by suppressing AP2A1, the results were stunning: the aged cells shrank back to their youthful size, cleared out accumulated waste, and resumed normal division.
How the Mechanism Works: The Integrin Connection
The researchers uncovered a sophisticated logistical failure that occurs as we age. Normally, AP2A1 helps cells move materials across their membranes (endocytosis). However, in aging cells, it gets hijacked. It begins transporting a sticky protein called integrin β1 directly to the cell's anchors (focal adhesions). This process acts like reinforcing superglue, causing the cells to flatten out, enlarge, and become immobile—a hallmark of the "zombie" cell state.
"We still didn't understand how these senescent cells can maintain their huge size," explained Pirawan Chantachotikul in a statement released alongside the findings. "One intriguing clue is that stress fibers are much thicker in senescent cells than in young cells." The team proved that AP2A1 is the architect of this thickening. By cutting the supply line (suppressing AP2A1), the cellular rejuvenation breakthrough forces the cell to dismantle its "old age" architecture and rebuild a youthful structure.
Why "Big" Cells Are a Big Problem
In the context of healthy aging science news, cell size is often overlooked, but it is critical. Senescent cells are abnormally large, which disrupts tissue organization and function. This hypertrophy is linked to chronic inflammation and metabolic dysfunction. The Osaka team's discovery that AP2A1 suppression restores normal cell size suggests that we can do more than just kill senescent cells (senolytics); we might be able to rehabilitate them.
Implications for Anti-Aging Medicine in 2026
This longevity protein discovery opens a new frontier for medical intervention. Unlike previous approaches that focused solely on clearing out dead wood, this method suggests a path to reversing senescence in humans by restoring the metabolic and structural integrity of existing cells. If drugs can be developed to safely inhibit the abnormal accumulation of AP2A1 on stress fibers without disrupting its vital functions elsewhere, we could see a new class of "senomorphics"—drugs that revert old cells to young ones.
"Our findings suggest that senescent cells maintain their large size through improved adhesion to the extracellular matrix," noted Professor Deguchi. This implies that by loosening this adhesion via AP2A1 targeting, we can physically unstick the jamming signals of aging. This approach could be revolutionary for treating age-related diseases such as fibrosis, cardiovascular decline, and skin aging.
A New Era of Rejuvenation
As the scientific community digests these findings, the focus is shifting rapidly toward clinical applications. With anti-aging medical breakthroughs accelerating, the identification of AP2A1 provides a concrete, druggable target. While human trials are the next logical step, the ability to turn back the clock in human cell cultures offers a promising proof of concept. For now, the AP2A1 protein stands as the most compelling new candidate in the global race to solve the problem of biological aging.