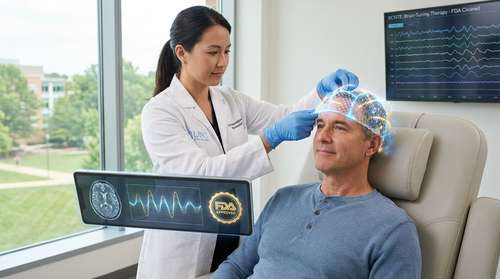
In a landmark decision that signals a new era for neurotechnology in 2026, the U.S. Food and Drug Administration (FDA) has officially approved XCSITE, a revolutionary noninvasive brain stimulation device developed by researchers at the University of North Carolina at Chapel Hill. This clearance marks a pivotal moment for mental health care, introducing a drug-free depression relief option that uses a personalized "closed-loop" mesh to synchronize disrupted brainwaves.
A New Standard in Noninvasive Mental Health Treatment
The approval, announced late Friday, centers on the XCSITE system, a wearable device that resembles a high-tech mesh cap. Unlike traditional brain stimulation methods that deliver a constant, one-size-fits-all electrical current, XCSITE creates a real-time feedback loop with the patient's brain. Developed under the leadership of Dr. Flavio Fröhlich at the UNC Chapel Hill neuroscience breakthrough lab, the device utilizes integrated EEG sensors to monitor neural activity and instantly adjust its stimulation to "retune" irregular rhythms associated with Major Depressive Disorder (MDD).
"This is not just about stimulating the brain; it is about conversing with it," said Dr. Fröhlich, director of the Carolina Center for Neurostimulation. "We have moved beyond the blunt instruments of the past to a precision therapy that listens to the brain's electrical language and helps it find its rhythm again." This capability positions XCSITE as a leader in the rapidly expanding field of neurotechnology 2026.
How Brainwave Synchronization Therapy Works
The core innovation behind XCSITE is its use of transcranial alternating current stimulation (tACS) in a closed-loop system. While earlier devices like tDCS (transcranial direct current stimulation) applied a static charge, XCSITE's brainwave synchronization therapy dynamic approach targets the specific alpha oscillations often disrupted in patients with depression. The mesh cap detects these desynchronized patterns and delivers imperceptible electrical pulses to guide neurons back into a healthy, synchronized state.
Dr. Fröhlich compares the process to an orchestra conductor bringing out-of-sync musicians back into tempo. "If the brain is an orchestra, depression is a state of dissonance," he explained during the press briefing. "XCSITE acts as the conductor, providing a subtle rhythmic cue that allows the brain to harmonize itself." This "brain-tuning" methodology minimizes side effects, avoiding the systemic issues often caused by pharmaceutical antidepressants.
Clinical Success and Drug-Free Depression Relief
The FDA's decision follows impressive results from multicenter clinical trials concluding in late 2025. Data showed that approximately 80% of participants experienced significant symptom remission after just two weeks of daily sessions. Crucially, the effects were sustained in follow-up assessments, suggesting that the device induces neuroplastic changes that help the brain maintain its healthy rhythm long after the device is removed.
Breaking the Treatment Resistance Cycle
For the millions of Americans suffering from treatment-resistant depression, XCSITE offers a lifeline. Traditional pharmacotherapy fails for nearly one-third of patients, leaving a massive gap in care. As a verified FDA approved depression device, XCSITE provides a viable alternative for those who have not responded to SSRIs or who cannot tolerate their side effects. The device's safety profile is pristine, with users reporting little to no sensation during the "tuning" sessions.
Availability and Future Implications
Pulvinar Neuro, the UNC spinoff company behind the technology now operating under Electromedical Products International, has confirmed that the XCSITE system will begin shipping to specialized psychiatric clinics by April 2026. A prescription-based home-use version is also in the pipeline for late 2027.
As the medical community digests this news, the implications extend far beyond depression. Researchers are already exploring XCSITE's potential for treating anxiety, chronic pain, and cognitive decline. But for now, the arrival of this XCITE brain stimulation technology represents a tangible hope for millions—a chance to retune their minds and reclaim their lives without a single pill.