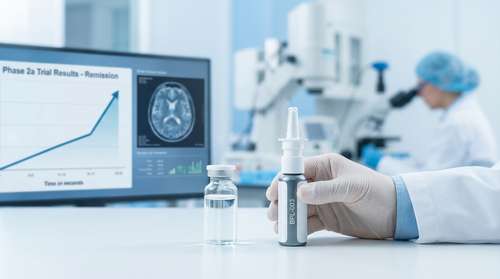
The landscape of psychiatric medicine shifted dramatically on March 18, 2026, with the publication of unprecedented clinical data. For millions of Americans battling intractable mood disorders, the experimental BPL-003 nasal spray offers a potent new beacon of hope. Developed as an intranasal formulation of the powerful psychedelic compound 5-MeO-DMT, this therapeutic intervention is breaking records in its clinical timelines. According to the newly released Phase 2a study results, the drug produced astonishing outcomes for patients diagnosed with treatment-resistant depression. A staggering 63% of trial participants achieved full clinical remission within an exceptionally short 90-minute window, a milestone that could entirely redefine standard care protocols for severe mental illnesses.
Unlike daily oral medications that can take weeks to build efficacy, this single-dose administration offers immediate intervention. Beyond the initial rapid onset, the long-term 5-MeO-DMT depression results demonstrated remarkable durability. Researchers reported that over 54% of treated patients maintained a robust clinical response for a full three months after receiving just one dose.
The Mechanics Behind Beckley Psytech BPL-003
To understand why this treatment-resistant depression breakthrough 2026 is capturing the attention of the global medical community, you have to examine the compound's unique pharmacokinetic profile. Beckley Psytech BPL-003 is a synthetic, patent-protected intranasal dry powder formulation of mebufotenin. Historically, naturally occurring psychedelics presented significant logistical hurdles in a clinical setting. Classic compounds like psilocybin or MDMA trigger intense, multi-hour hallucinogenic effects, which require extensive facility resources, overnight stays, and prolonged medical supervision.
BPL-003 completely subverts this traditional paradigm. The proprietary nasal delivery system ensures rapid absorption directly across the mucous membranes, triggering a profound therapeutic effect that peaks and resolves quickly. Because of this short-acting mechanism, patients in the Phase 2a trial were consistently ready for clinical discharge in under two hours. This practical 90-minute clinical window mirrors the workflow already established by approved interventional psychiatric treatments, making it highly scalable for existing mental health clinics and hospital networks.
Analyzing the Unprecedented Phase 2a Data
When evaluating psychedelic clinical trials FDA regulators prioritize two primary metrics: absolute patient safety and measurable efficacy. The dataset published this week provides compelling, quantified evidence on both fronts. Utilizing the Montgomery-Åsberg Depression Rating Scale (MADRS)—the gold standard for measuring depression severity—investigators closely tracked patient progress from baseline through the critical 12-week post-dosing period.
- Rapid Onset: Within the first 90 minutes of administration, 63% of patients hit the threshold for clinical remission. They reported an almost immediate lifting of the heavy, paralyzing symptoms that had previously resisted multiple standard treatments.
- Sustained Efficacy: The neuroplasticity triggered by the acute psychedelic experience appears to yield lasting structural and psychological benefits. Maintaining a 54% clinical response rate at the three-month mark from a single dose is practically unheard of in modern psychopharmacology.
- Safety Profile: The treatment was exceptionally well-tolerated. Adverse events were predominantly mild to moderate—such as temporary headaches or mild nausea—and resolved quickly without targeted medical intervention. Most importantly, no serious adverse events were recorded during the trial period.
A Major Milestone for Fast-Acting Antidepressant Therapy
Standard selective serotonin reuptake inhibitors (SSRIs) fail roughly one-third of the patients who try them. For these individuals, the diagnostic label of treatment-resistant depression often translates into a grueling lifetime of trial-and-error pharmacology. They endure compounding side effects with little to no psychological relief. The introduction of a reliable, fast-acting antidepressant therapy could drastically alter this bleak trajectory, offering a functional reset for the brain's default mode network.
While patients receive psychological support—including preparation, dosing supervision, and integration sessions—the true driver of this rapid recovery is the compound's ability to facilitate immediate cognitive flexibility. Patients stuck in rigid, negative thought loops are suddenly granted the mental bandwidth to break free and rebuild healthier neural pathways.
What This Means for Mental Health Medical News
As the psychiatric field digests this major mental health medical news, attention is naturally pivoting toward the next regulatory hurdles. The impressive safety and efficacy data from this open-label Phase 2a trial provide a robust, scientifically validated foundation for upcoming end-of-Phase 2 meetings with regulatory bodies. By proving that a short-acting psychedelic can safely deliver long-term relief, researchers are successfully dismantling the logistical barriers that have historically plagued psychedelic medicine.
The journey from successful Phase 2a trials to widespread commercial availability requires further validation. Larger, quadruple-masked, randomized Phase 2b and Phase 3 trials are already actively enrolling to confirm these initial findings across broader, more diverse patient populations.
If these subsequent trials mirror the remarkable outcomes published this week, the psychiatric community could soon have access to a highly scalable, single-dose intervention. Until then, practitioners and patients alike are watching closely, recognizing that this latest development represents one of the most significant and actionable strides forward in depression treatment in decades.